 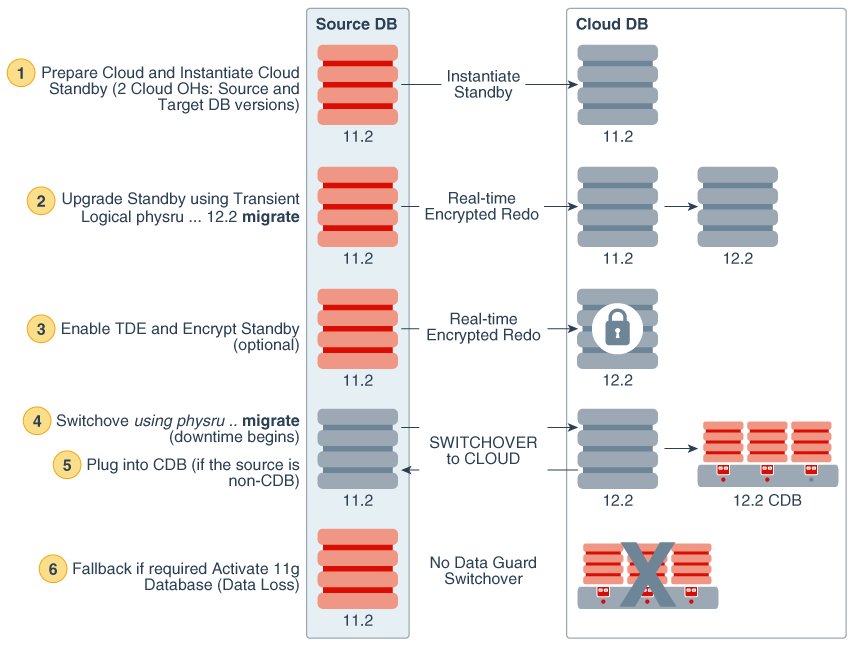
With InForm Advanced Molecular Analytics, researchers have consolidated access to both clinical and genomic data for numerous purposes such as submission or the design of more exploratory translational research studies.Ĭreate competitive clinical differentiation messages for new drugs in highly competitive markets where comparative effectiveness, outcomes, clinical utility and cost-effectiveness are critical factors to ensure commercial success.Įnable clinical researchers to study patient genomic profiles, analyze differences and compare molecular data against common large public domain data sets, increasing statistical power. Reduce or eliminate the difficult manual methods to collect and integrate data into a single database for prospective analysis and faster decision-making. 
The solution also helps researchers explore synergistic programs through post-trial, cross-study analysis with combined molecular and clinical data. Stratify patients and reduce research and development costs and time by leveraging targeted drug development approaches and decreasing study size and length. Oracle Health Sciences InForm AMA helps life sciences organizations to: The solution leverages proven technology from the Oracle Health Sciences cloud-based Translation Research Center to deliver the first “molecular-aware” clinical data capture and management solution. Leveraging this seamless process enables improved trial efficiency by reducing the sample sizes and potentially the treatment duration required to demonstrate statistical and clinical outcomes. Oracle Health Sciences InForm AMA facilitates the prospective dynamic randomization of patients with or without “omics” signatures into respective treatment arms to optimize study conduct. The solution is integrated with Oracle Health Sciences InForm EDC platform, letting clinical researchers effectively capture, aggregate and analyze molecular data along with clinical data, providing the information needed to identify new predictive biomarkers. Oracle Health Sciences InForm Advanced Molecular Analytics (AMA) is the first commercially available cloud-delivery solution to incorporate genomics into the clinical data capture and management process. The use of prospective genomic testing can optimize patient inclusion into clinical trials, ensuring enrollment of patients that have the best chance of demonstrating a positive therapeutic effect. 
Pressing Need for Managing Biomarker Data In Clinical Trials As the industry moves aggressively forward to expand the effective application of personalized medicine, biomarkers are becoming a key component of clinical development programs. 
The solution extends the capabilities of Oracle Health Sciences InForm, the industry-leading clinical data capture and management solution to effectively collect, store and analyze genomic data in clinical trials. Oracle Health Sciences InForm Advanced Molecular Analytics enables life sciences organizations to incorporate genomic data into clinical R&D, enabling more targeted, biomarker-driven clinical trials to optimize trial outcomes and increase efficiency. (SaaS) and fully integrated with Oracle Health Sciences InForm Cloud Service Identify potential risks to trial timelinesĭomain data for joint analysis with your own data.Utilize public domain data to draw and compare insights, resulting in faster identification of targets With correlation analysis between clinical outcomes and molecular biomarkers Study cycle-times and accelerating time to launch Ĭomprehensive 21CRF-Part11compliant solution for data management and analysis ORACLE HEALTH SCIENCES INFORM ADVANCED MOLECULAR ANALYTICS INCORPORATE GENOMIC DATA INTO CLINICAL R&D 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
